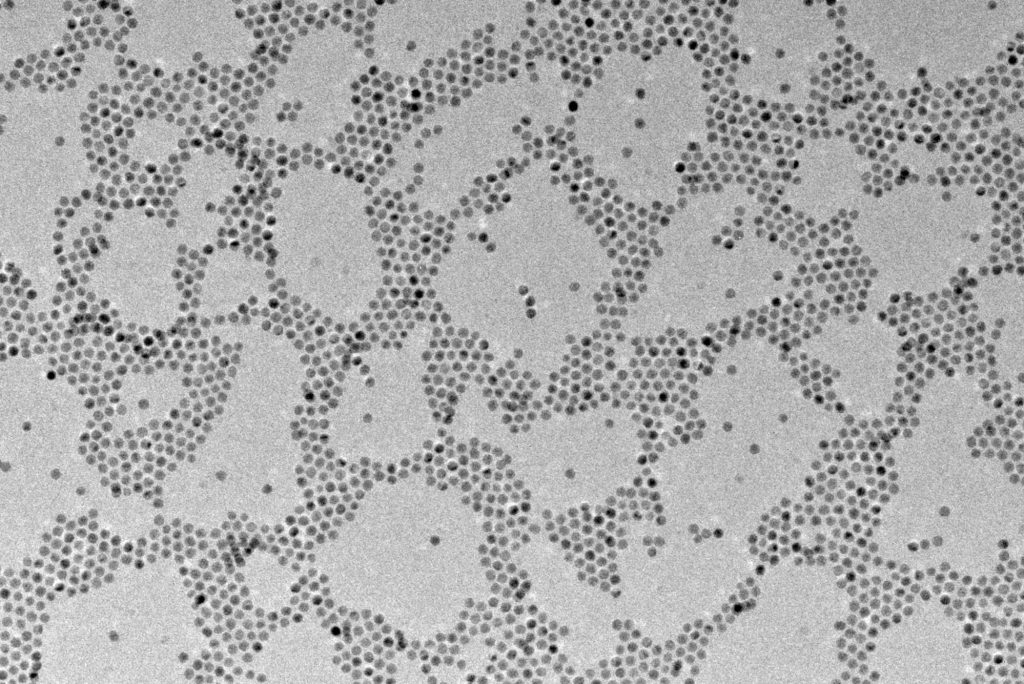
What are nanoparticles and what are they used for?
A nanoparticle is defined as a particle of matter that is between 1 and 100 nanometers in diameter. Due to their submicroscopic size, nanoparticles display a range of unique material properties (large surface area/volume ratio, quantum mechanical effects etc), offering a wide range of advantages over regular materials in fields such as medicine, agriculture, engineering, electronics etc.
Due to their novel properties, the ability to synthesise nanoparticles with good and controlled qualities is desirable in various fields of application – with the method of synthesis falling into one of two general strategies.
- Top-down – breakdown or leaching of nanoparticles from bulk material
- E.g. Milling, ion/plasma etching, lithography
- Bottom-up – coallesence or assembly of atoms/molecules into nanoparticles
- E.g. Chemical precipitation, CVD, polymerization
Nanoparticle properties are closely linked to their size, shape, composition and surface structure – therefore accurate control of reaction conditions is key to ensuring nanoparticles with the desired properties. Key synthesis parameters include temperature, mixing, reagent concentrations and reaction time.
Nanoparticle synthesis in batch
Typically, nanoparticle synthesis in batch reactors focus on “wet chemical” processes to form an insoluble precipitate of the desired nanoparticles. The resultant particles are then separated from the solvent by a combination of filtration, centrifugation, washing, sedimentation and evaporation.
Batch processing of nanoparticles is a cost effective and convenient method for nanoparticle synthesis and allows for fine control of the particle’s chemical composition – however care must be taken to ensure batch-to-batch consistency of nanoparticle properties.
Jacketed lab reactors provide an excellent solution for nanoparticle synthesis, offering excellent mixing and pin-point temperature control, as well as the ability to automate your entire procedure (including dosing protocols) for the highest levels of reproducibility.
Multiple sensor can also be used to accurately monitor and control all reaction parameters (temperature, pH, turbidity etc) in real time.
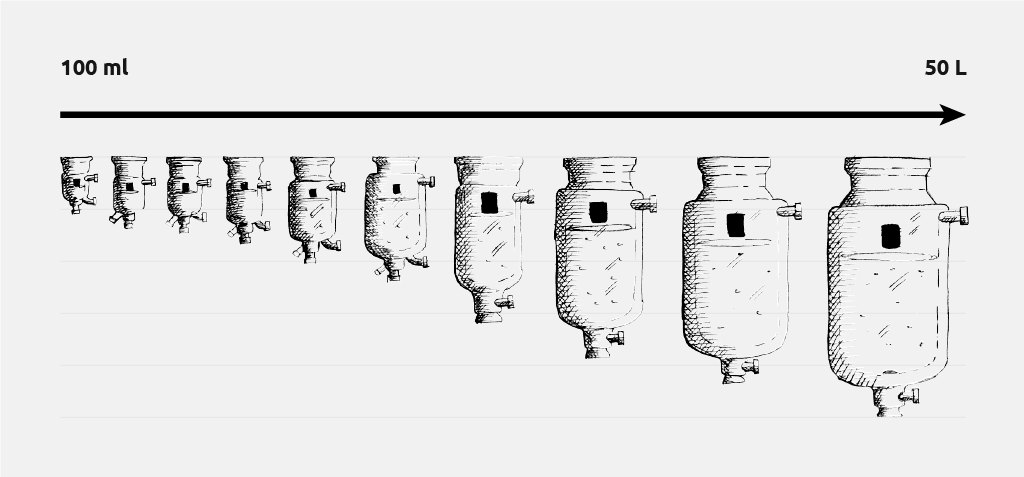
Jacketed lab reactors also simplify the process of scaling nanoparticle synthesis, by utilising common reactor geometries across a large volume range found at manufacturing scale.
How can Syrris help chemists performing nanochemistry?
Syrris offers a wide range of innovative batch and flow reactors for nanoparticle synthesis. The award-winning systems, designed by chemists for chemists, present multiple benefits:
- Narrow particle size distribution: Excellent mixing and temperature control leads to improved homogeneity of particle size
- Rapid nanoparticle optimization: Process conditions such as temperature, time, mixing, reagent ratios and concentrations can be quickly varied
- Reproducibility and automation: Additions, mixing, temperature, etc. can be fully controlled in an automated fashion for seamless reproducibility
- Easy to use: All products can be assembled, maintained and cleaned without tools
- Simple scale up: Large range of reactor sizes (batch) and flow rates (continuous flow) provides the tools for moving from small-scale process optimization to production
- Nanoparticles not possible by other means: Nanoparticles previously unseen using batch techniques can be obtained in microreactors due to the high level of control
The list below shows a selection of nanoparticles synthesized in Syrris reactor systems and the unique advantages they have offered:
| Nanoparticle Synthesised | Unique advantage using Syrris reactor systems |
| Nickel nanocubes | Ability to accurately select the desired shape. High-quality material obtained |
| Silver nanowires | Excellent monodispersity. Improved electric conductivity property |
| Gold nanocrystals | Smaller average particle size obtained in flow. Easy process optimization |
| Silica beads | Narrow size distribution. High reproducibility |
| Platinum nanoparticles deposited on carbon fibers | Synthesis and deposition performed in one cycle. Control of the size by varying flow rate |
| Cadmium selenide quantum dots | Synthesis, capping, and functionalization is done in one continuous process. Large-scale synthesis |
| Superparamagnetic iron nanoparticles | Small particle size. Fast throughput |
| Nickel tetrapods | Previously unreported shape. Only seen in microreactors |
| Titanium oxide nanoparticles | Faster synthesis. Excellent yields |
| Amino-functionalized graphene quantum dots couple with gold nanoparticles | Coupled amino-functionalised graphene quantum dots to gold nanoparticles in batch. |
| Iron nanoparticles | Large scale batch synthesis. Atlas pump used for reagent dosing. |
| Magnetic iron nanoparticles | Used externally mounted magnetic rods for particle separation |
Nanoparticle synthesis customer stories
Reproducible scaling of Gold nanoparticles in Brazil
Researchers at a University in Brazil have been using the Orb Jacketed Reactor system to scale up their gold nanoparticle synthesis. Prof. Ricardo Aucélio from Pontificia Universidade Católica research looks at specialist nanoparticles for use in analytical tools. They purchased the Orb jacketed reactor system in an attempt to overcome the scaling issues of moving from a round bottom flask.
The Orb system is very simple to operate, compact and robust” says Prof. Aucelio. “We are using it for the synthesis of gold nanoparticles (AuNPs) capped with different chemical binders. As the system is very easy to operate, my masters´ and doctoral students already have full autonomy to use the system.”
Midatech use Atlas reactors to produce gold nanoparticles for pharmaceutical applications
Midatech Biogune, a Spanish biotechnology company, are using Syrris Atlas HD automated jacketed reactor systems to produce custom-made functionalized gold nanoparticles for medicinal use.
The Atlas HD Potassium was the only logical way to move from manufacturing nanoparticles on a laboratory scale to a mini production environment.”
Nanoparticle synthesis publications
Syrris products have been used for the synthesis and study of various nanoparticles including:
Nanoparticle synthesis in batch chemistry publications:
-
Large-Scale Synthesis of Colloidal Fe3O4 Nanoparticles Exhibiting High Heating Efficiency in Magnetic Hyperthermia
First published: 2nd April 2014 | https://doi.org/10.1021/jp500816u
This paper used Atlas HD automated jacketed reactor to synthesise iron nanoparticles on large scale, using Atlas syringe pump to dose reagents.
-
Determination of Aflatoxin M1 in Milk by a Magnetic Nanoparticle-Based Fluorescent Immunoassay
First published: 17 Jan 2017 | https://doi.org/10.1080/00032719.2016.1187626
This paper used Globe jacketed lab reactor for the preparation and functionalisation of magnetic nanoparticles.
-
Preparation of Paraffin-Based Solid Combustible for Hybrid Propulsion Rocket Motor
First published: 02 Dec 2016 | https://doi.org/10.2514/1.B36197
This paper used Atlas HD automated jacketed reactor to create paraffin particles with a controlled range of diameters.
-
Unexpected Secondary Nucleation in Poly(Vinyl acetate) Nanoparticle Synthesis by Ab Initio Batch Emulsion Polymerization Using Poly(Vinyl alcohol) as Surfactant
First published: 13th Aug 2019 | https://doi.org/10.1002/mren.201900024
This paper used Atlas HD automated jacketed reactor for emulsion polymerisation to produce polymer nanoparticles.
Nanoparticle synthesis in flow chemistry publications:
-
Green microfluidic synthesis of monodisperse silver nanoparticles via genetic algorithm optimization
Using the Syrris Asia flow chemistry system, researchers at the Department of Chemistry (Uppsala University, Sweden) have reported the scalable and green synthesis of monodisperse silver nanoparticles via genetic algorithm optimization.
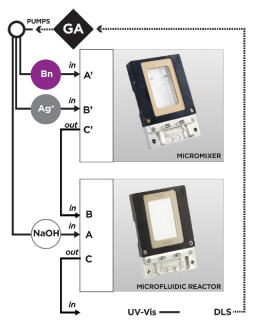
First published: 23rd July 2015 | https://doi.org/10.1039/C6RA20877K
This publication uses an Asia flow chemistry system.
-
Sensitive progesterone determination using a magnetic particle-based enzyme-linked immunosorbent assay
This publication describes the synthesis, silane functionalization and use of magnetic Iron nanoparticles in combination with enzyme-linked immunosorbent assay (ELISA) to facilitate the hybrid technique of ‘magnetic nanoparticles – ELISA’ for use in the capture of progesterone in numerous types of milk. The synthesis and functionalization of the magnetic nanoparticles were carried out in an automated Globe system (now replaced by the Syrris Orb Jacketed Reactor) to furnish nanoparticles with a mean size of 182.4nm and 292.8nm after functionalization.
First published online: 24th November 2014 | https://doi.org/10.1556/JFC-D-14-00009
-
Nickel nanocrystals – fast synthesis of cubes, pyramids, and tetrapod
In this paper, the authors study the formation and growth of nickel nanocrystals/nanoparticles using organometallic Ni(?4-C8H12)2 as a precursor. The reaction temperature has been identified as the key parameter which determines the shape of the obtained nanoparticles. In addition to the three classic shapes of nanoparticles (cubical, spherical, pyramidal), a new shape of nanoparticles – tetrapods – has been reported. This shape has only been observed in Syrris microreactors because of the excellent heat transfer and temperature control obtained in those devices.


First published: 12st November 2012 | https://doi.org/10.1039/C2RA22024
This paper uses an Asia flow chemistry syringe pump and a glass microreactor.